Symetrian™ is a standardized, patent pending, Aloe-based composition for a healthy immune homeostasis.* It is clinically proven to promote rapid immune cell activation and production of circulating TCRγδ+ Gamma delta T cells, suggestive of its heightened immune surveillance at portals of entry, providing immediate immune response.*
Symetrian™ has also been clinically proven to increase the levels of glutathione peroxidase (GSH-Px) highlighting its strong antioxidant activity.* Symetrian™ is composed of plant extracts standardized for specific polysaccharides and polyphenols from three botanicals with a long history of safe human consumption for immune support, Aloe barbadensis (Aloe vera), Poria cocos and Rosemary (Rosmarinus officinalis).
What Makes Symetrian™ Unique?
- Standardized, patent-pending, novel Aloe-based composition with long history of safe human consumption for immune support*
- Suitable as a long-term daily supplement for all populations.
- Extensive scientific evidence including two randomized, double-blind placebo-controlled, IRB approved clinical studies
- Clinically proven to support maintenance of healthy immunity homeostasis*
- Clinically proven to support cellular and humoral immunity by increasing TCRγδ+ Gamma delta T cells and IgG antibodies*
- Clinically proven to support healthy inflammatory response by maintaining healthy cytokine levels and cytokine responses*
- Clinically proven to support strong antioxidant activity*
- Clinically proven to support rapid immune activation and response*
- Clinically proven to activate Natural killer cells and Gamma delta T cells within hours*
Plant Origin
Derived from Aloe vera, Poria cocos, and Rosemary extracts.
Applications
- Maintenance of immunity homeostasis*
- Provide heightened mucosal immunity at the portal of entry for increased immune surveillance and/or a robust response*
- Promote innate and adaptive immunity*
- Provide strong antioxidant activity*
Formulation
Can be used as an active agent alone or formulated with other cold/flu agents in tablets, capsules, gummi, gels, liquids, powders, bars and other delivery systems.
Physical Properties
Brown colored powder easily suspended in water.
*Indications and claims related to the health benefits or property of an ingredient or product are intended for industry only and are governed in accordance with country-specific laws and regulations and may not be appropriate for final consumer products. In the United States, it is your responsibility to ensure that product claims and indications are in compliance with all applicable laws and regulations, including the Federal FD&C Act and the FTC Act. In all other countries, please consult with a local regulatory or legal professional who may provide you with competent advice and guidance.
Discovery
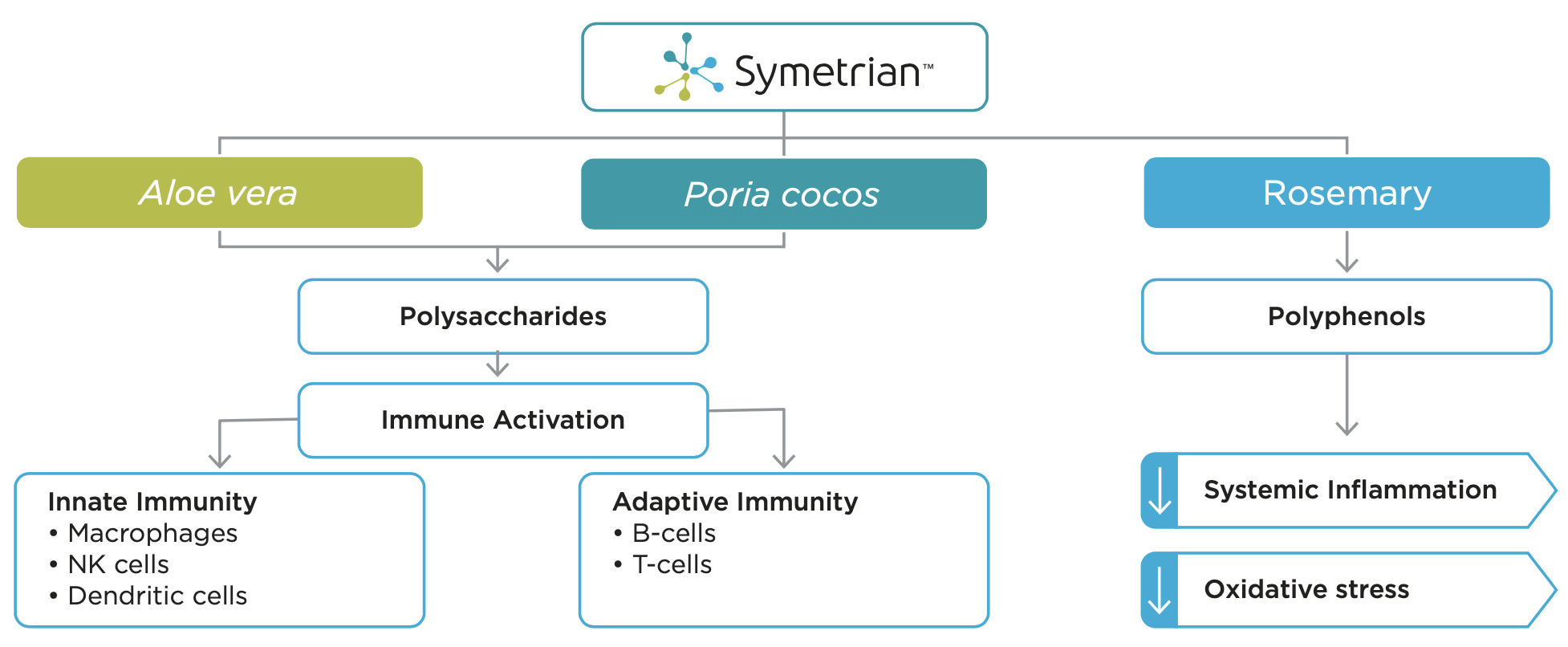
Symetrian™’s unique formulation was developed through Unigen’s proprietary Phytologix® technology platform, based on a collection of more than 12,000 plants and marine samples with documented human consumption. Unigen scientists used this library of comprehensive botanical profiles to identify plants that delivered immune support benefits. This is the first instance of historically safe medicinal plants, Aloe barbadensis (Aloe vera), Poria cocos and Rosmarinus officinalis (Rosemary) being standardized in such a way to achieve immune homeostasis. The actives in these botanical extracts contribute clinically meaningful mechanisms of action to provide Symetrian™ its immune modulation characteristic, suggesting its usage for continual immune readiness and surveillance purposes.Mechanism of Action
Immune homeostasis and/or immune stimulation and modulation of Symetrian™ is derived from:- Support of the body’s innate immunity
- Promotion of the body’s natural adaptive immunity
- Support of the body’s natural responses to inflammation and oxidative stress
Scientific Research
Pre-Clinical Data
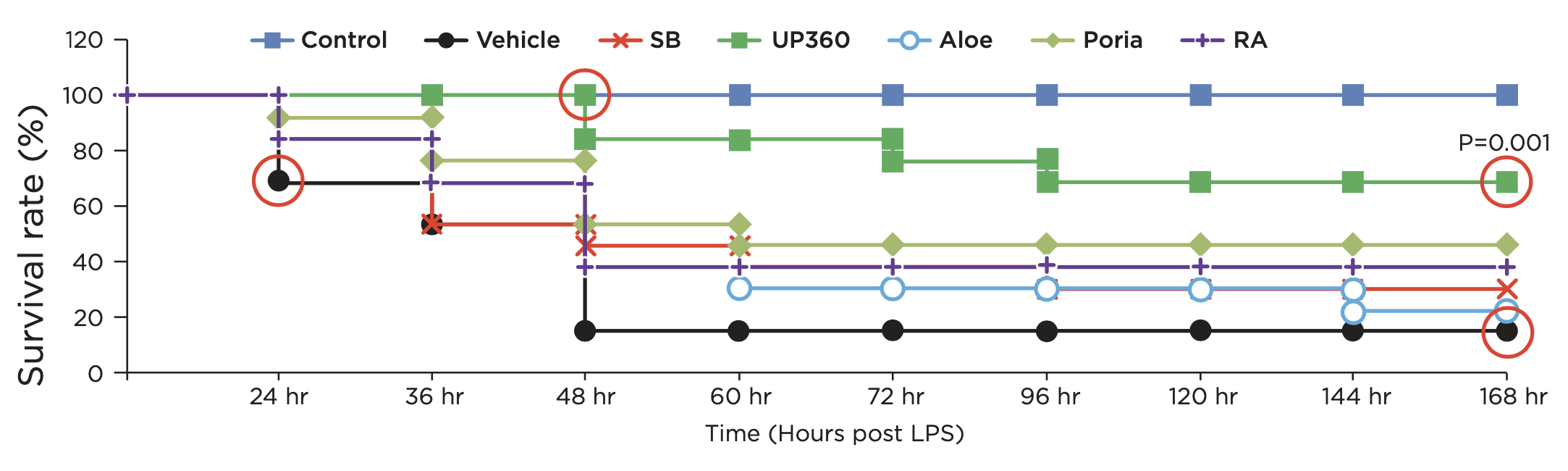
Pre-Clinical Data on Acute Lung Injury and Inflammaging Induced Immune Senescence- Symetrian™ protected animals from Lipopolysaccharide (LPS)-induced sepsis (Figure 2)
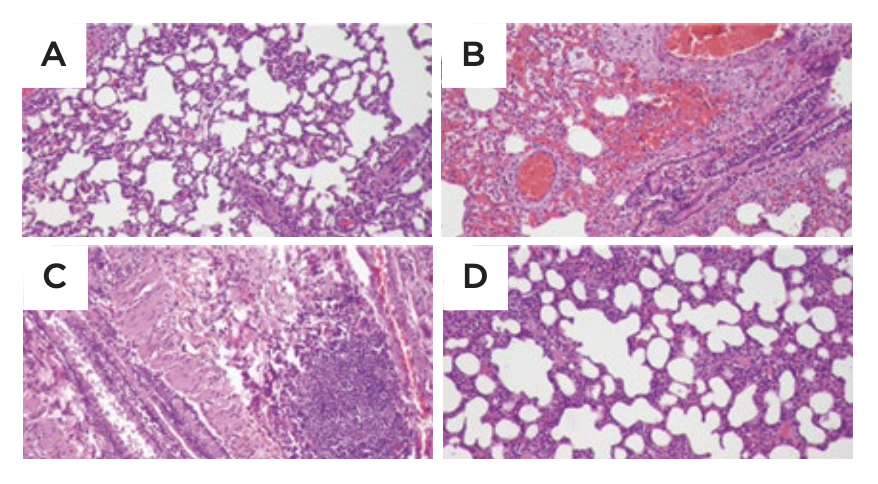
- Symetrian™ supported health of lungs (Figure 3) in LPS-induced acute lung model, with reduction of key cytokines, chemokines (TNF-α, IL-6, IL-1, CRP, CINC-3)
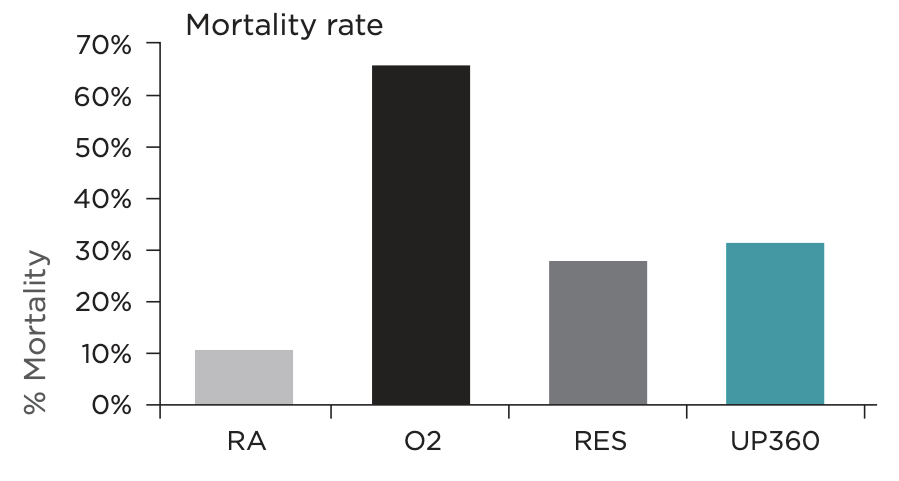
- Symetrian™ increased survival rate in hyperoxia-induced oxidative stress mouse model (Figure 4)
- Symetrian™ decreased bacterial load in the broncho-alveolar lavage in hyperoxia-induced oxidative stress mouse model, highlighting its ability in clearing airway bacteria in the lung (Figure 5)
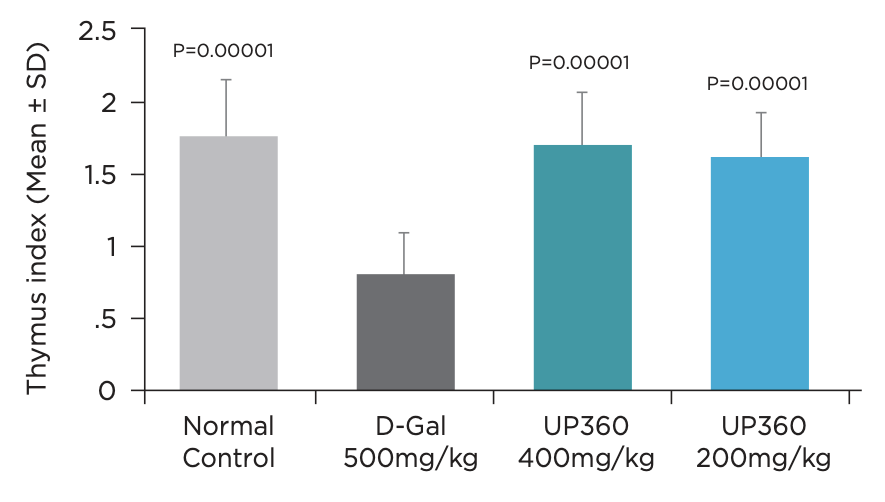
- Symetrian™ supported the body’s natural response to protect thymus against oxidative stress-induced immune senescence, which may affect the body’s ability to mount an immune response (Figure 6)
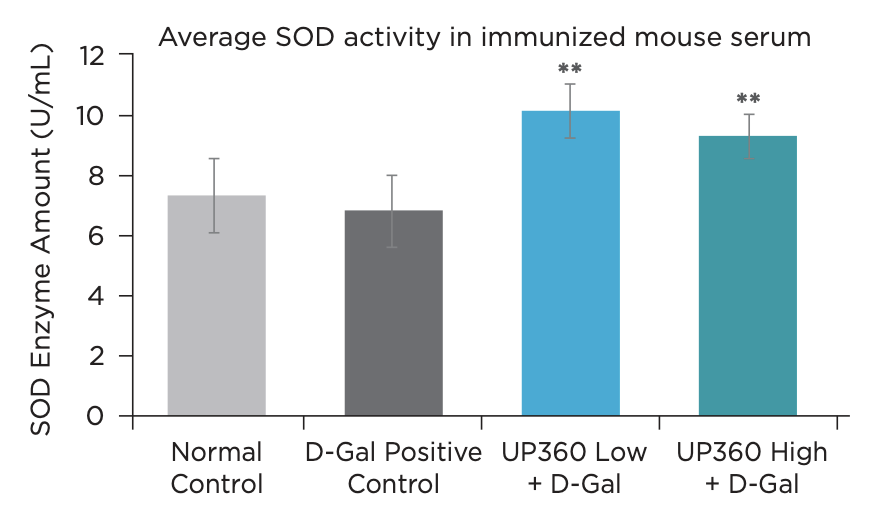
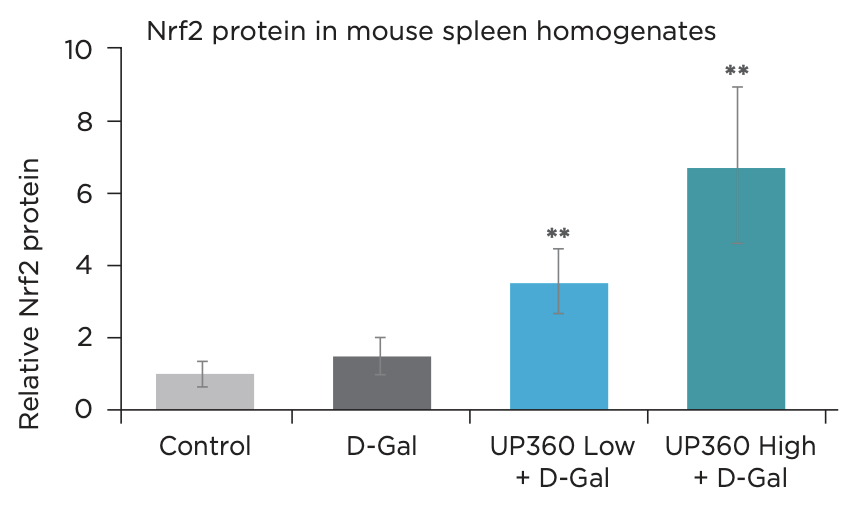
- Symetrian™ supported increased SOD activity and increased anti-oxidative protein NRF2, indicating an increased capacity to neutralize free radicals (Figure 7 & 8)
- Symetrian™ supported increased CD3+ T-cells, CD4+ helper T-cell, CD8+ Cytotoxic T cells, NKp46+ Natural Killer cells, CD4+TCRγδ+ Gamma delta T cell levels, IgA, demonstrating that the composition primes the immune system and causes expansion of innate and adaptive immune cell populations (Figure 9)

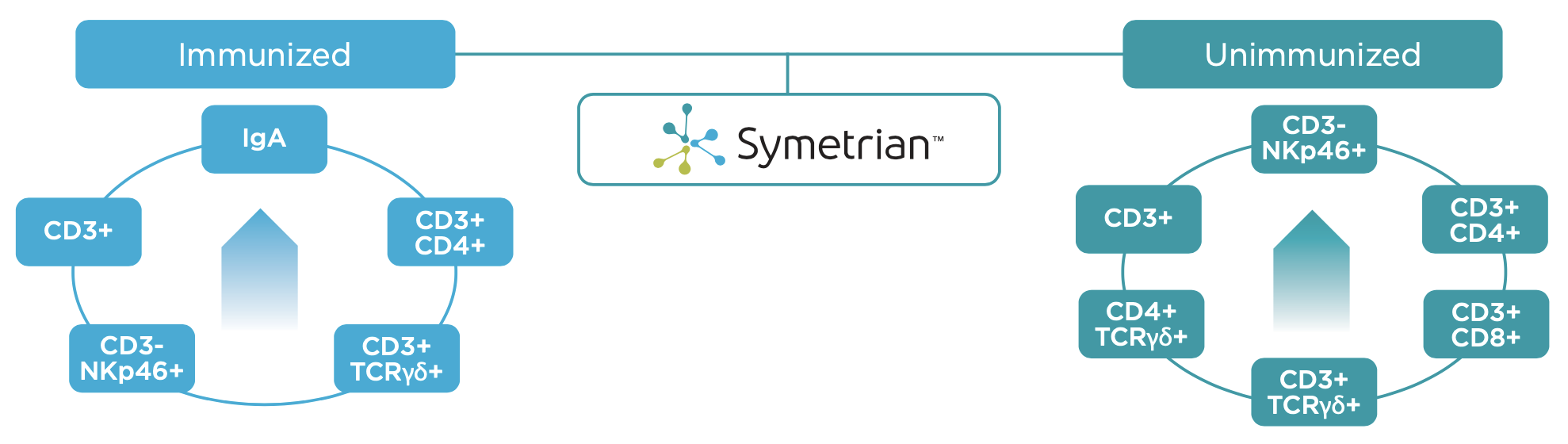
Clinical Data
Clinical Data on Immune Support
- Supplementation with Symetrian™ 500 mg twice per day was found to increase expansion of Gamma Delta (γδ) T Cells (Figure 10) suggesting a more robust mucosal cellular response highlighting its primarily role in first line of defense and immune surveillance
- Symetrian™ supplementation showed statistically significant increase in influenza B-specific IgG levels, in the post-vaccination period indicating enhanced support of humoral protective immunity
- Supplementation with Symetrian™ supported increased serum glutathione peroxidase (GSH-Px) levels highlighting improved antioxidation capacity
- Symetrian™ supplementation produced statistically significant increase in the level of serum IL-1RA, indicating its property maintaining healthy cytokine response
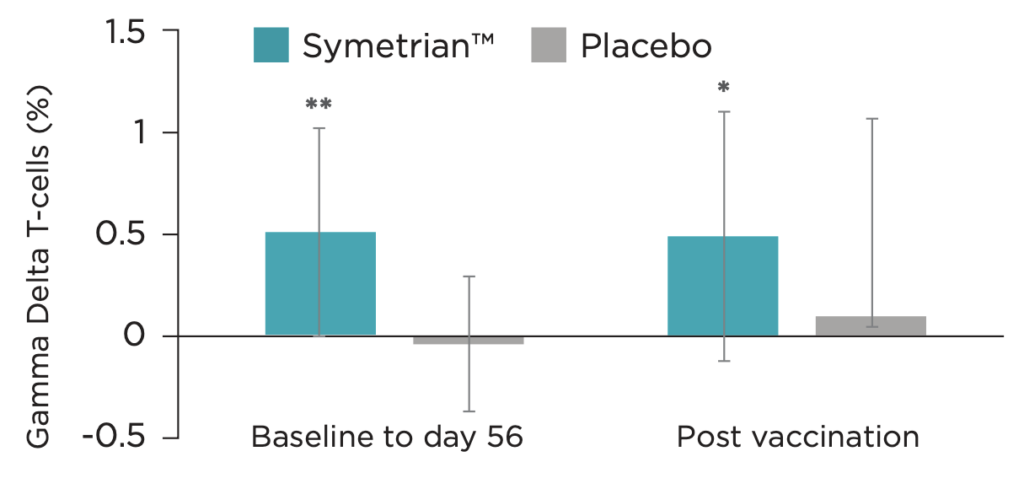
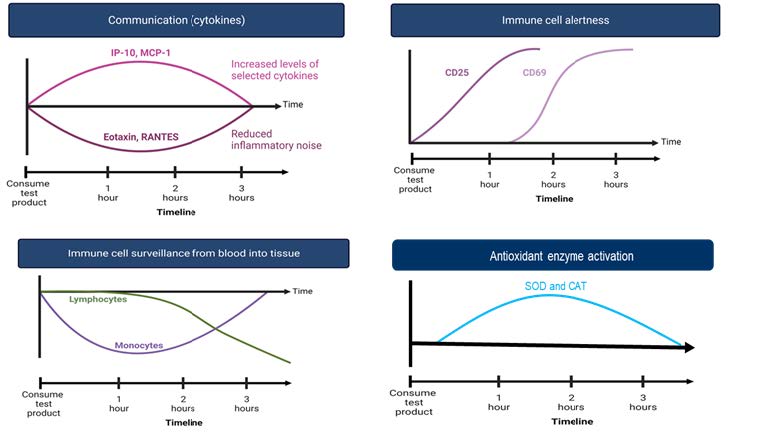
Clinical Data on Rapid Immune Modulation
- Supplementation with Symetrian™ at 500 mg single oral dose showed rapid immune activation and proliferation in a randomized double-blind placebo-controlled cross over human clinical study
- Supplementation with Symetrian™ showed progressive activation in the numbers of lymphocytes (NKT cells, NK cells; cytotoxic T cells, γδT cells) over a 3-hour period in the blood where the percent changes were found statistically significant against placebo.
- Supplementation with Symetrian™ increased activation markers (CD25, CD69 and CD56) on innate and adaptive immune cell populations (monocytes, NK cells, NKT cells, and γδT cells) as early as 1 hour post 500 mg oral supplementation. The CD25 increase after 1 hour and 2 hours was statistically significant for the NKT cells and γδT cells, respectively
Product Safety
Safety is a top priority at Unigen. All the ingredients we develop undergo rigorous safety testing using state-of-the-art in vitro, pre-clinical, and/or human safety studies (for some ingredients). Symetrian™ was found safe through these stringent safety studies.*References
- Beyer WE, Van der Logt JT, van Beek R, Masurel N. Immunoglobulin G, A, and M response to influenza vaccination in di erent age groups: e ects of priming and boosting. J Hyg (Lond). 1986 Jun;96(3):513-522.
- Sabbaghi A, Miri SM, Keshavarz M, Mahooti M, Zebardast A, Ghaemi A. Role of γδ T cells in controlling viral infections with a focus on influenza virus: implications for designing novel therapeutic approaches. Virology Journal 2020;17(1):174. doi: 10.1186/s12985-020-01449-0.
- Swarup V, Ghosh J, Ghosh S, Saxena A, Basu A. Antiviral and anti-inflammatory e ects of rosmarinic acid in an experimental murine model of Japanese encephalitis. Antimicrob Agents Chemother. 2007 Sep;51(9):3367-70.
- Novak M, Vetvicka V. Beta-glucans, history, and the present: immunomodulatory aspects and mechanisms of action. J Immunotoxicol. 2008 Jan;5(1):47-5
- Sierra-Garcia GD, Castro-Rios R, Gonzalez-Horta A, Lara-Arias J, Chavez-Montes A. Acemannan, an Extracted Polysaccharide from Aloe vera:A Literature Review. Nat Prod Commun. 2014;9:1217–21.
- Choi S, Chung MH. A Review on the Relationship between Aloe vera Components and their Biologic E ects. Semin Integr Med. 2003;1:53–62.
- Bulut O, Kilic G, Domínguez-Andrés J, Netea MG. Overcoming immune dysfunction in the elderly: trained immunity as a novel approach. Int Immunol.2020 Nov 23;32(12):741-753.
- Yimam M, Horm T, O’Neal A, Jiao P, Hong M, Jia Q. UP360, a Standardized Composition from Extracts of Aloe barbadense, Poria cocos, and Rosemary officinalis Protected Against Sepsis and Mitigated Acute Lung Injury in Murine Models. J Med Food 2023 Jul;26(7):489-499. doi: 10.1089/jmf.2022.0136. Epub 2023 May 16.
- Lewis ED, Crowley DC, Guthrie N, Evans M. Healthy adults supplemented with a nutraceutical formulation containing Aloe vera gel, rosemary and Poria cocos enhances the effect of influenza vaccination in a randomized, triple-blind, placebo-controlled trial. Front. Nutr., 24 April 2023 Sec. Nutritional Immunology Volume 10 – 2023
- Yu L, McGarry S, Cruickshank D, Jensen GS. Rapid increase in immune surveillance and expression of NKT and γδT cell activation markers after consuming a nutraceutical supplement containing Aloe vera gel, extracts of Poria cocos and rosemary. A randomized placebo-controlled cross-over trial. Plos One September 12, 2023
*These statements have not been evaluated by the Food & Drug Administration. This product is not intended to diagnose, treat, cure, or prevent any disease. These statements are presented for informational purposes only and are not intended to be presented to final consumers.
Want to learn more about Symetrian™?
"*" indicates required fields